Writing HSC Chemistry study notes might seem pretty simple, but once you sink you fangs into it it’s always a surprise to see how much there actually is to it!
On top of the differences between handwritten and typed notes, highlighters and coloured pens and loose-leaf paper versus bound books, every subject seems to want theirs written differently.
So to help you out with at least that last one, we’ve put together this handy guide to make effective HSC Chemistry study notes. Couple good note-taking skills with these Top 5 HSC Chemistry Study Tips and you’ll be that much closer to a Band 6!
Step 1: Target the syllabus
Step 2: Write them out!
Step 3: Research anything you don’t understand independently
Step 4: Look for connections between syllabus dot points
Step 5: Jump into practice questions
Step 1: Target the syllabus
Don’t know where to start?
The syllabus is a great place to get an idea of the learning outcomes that NESA wants you to master.
It’s good to check that you understand which content falls under which syllabus point by:
- Organising your notes under sub-headings derived from the syllabus
- Or labelling relevant syllabus dot points next to pieces of information in your note set.
Since some of the content in the new syllabus can be a bit confusing, don’t be afraid to ask your teacher or tutor for clarity and advice.
Let’s take this section of the syllabus for an example:
You could organise your notes like this:
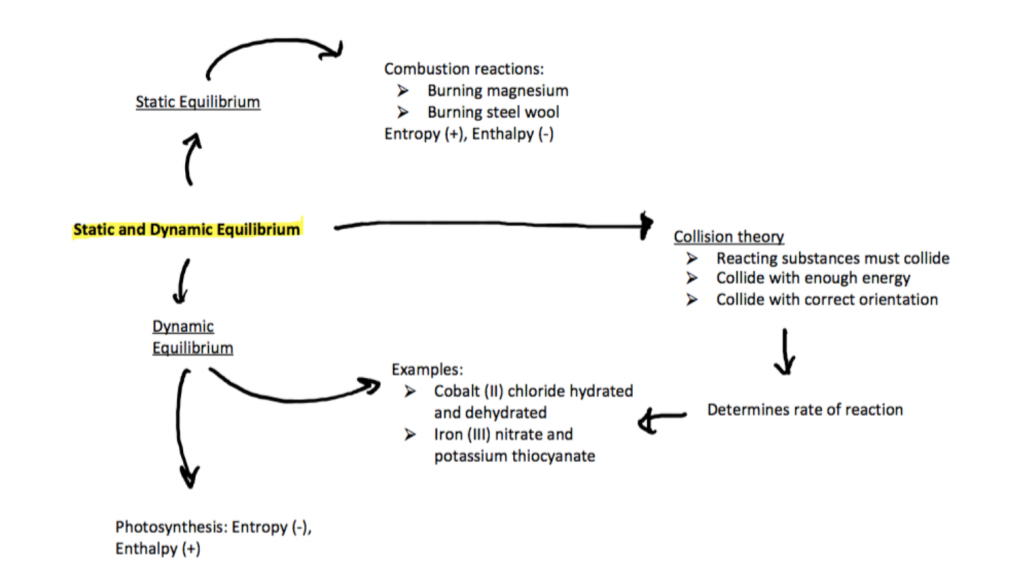
Point 5.1: Static and Dynamic Equilibrium – what happens when chemical reactions do not go through completion?
- Static equilibrium occurs when the system is at rest and the chemical reaction has stopped.
- Examples of static equilibrium can include burning magnesium and steel wool
- Dynamic equilibrium occurs during a reversible reaction whereby the rate of forward reaction is the same as the rate of the reverse reaction.
- Examples of dynamic equilibrium can include reaction between cobalt (II) chloride hydrated and dehydrated and iron (III) nitrate and potassium thiocyanate
Or like this:
- Static equilibrium occurs when the system is at rest and the chemical reaction has stopped (5.1.2).
- Examples of static equilibrium can include burning magnesium and steel wool (5.1.1, 5.1.2, 5.1.3)
- Dynamic equilibrium occurs during a reversible reaction whereby the rate of forward reaction is the same as the rate of the reverse reaction (5.1.2)
The most important thing is that you can see how the content relates to the syllabus! (Don’t feel pressured to write your notes in full sentences when you’re navigating the syllabus – you can stick to what you are comfortable with).
Want to structure your notes like this? We’ve done the hard work for you! Check out our HSC Smart Notion Template, which organises your HSC Chemistry notes by syllabus dot points so that it’s 100x easier to study for exams!
Step 2: Write them out!
Once you’ve decided which note taking style works best for you and understand what the syllabus wants you to learn, it’s time to write your HSC Chemistry study notes.
As Chemistry involves a lot of processes and concepts that relate to one another, try out diagrams, tables or mind maps to draw connections and develop that holistic thinking NESA is looking for. Sometimes, exams might even require you to use these formats so let your creativity run loose!
For example, you may draw a conceptual mindmap like this:
If you want to make digital notes, you can use these websites:
- Draw.io – for flowcharts or mind maps with proper scientific formatting
- Bubbl.us – for quick and easy visual guides with less stress
That said, chemistry notes are always easier to make by hand writing because of its complex chemical equations and diagrams. Hand writing notes also helps with remembering them so it’s like killing two birds with one stone yay!
Depending on how you are coping with the content, it’s good to write out your notes once a day or once a week by the topic area you have covered in class. For the more difficult concepts, try revisiting over the term to ensure you build a strong understanding!
Always falling behind on your study notes? Start using these 4 Efficient HSC Classroom Habits to see a massive difference!
Step 3: Research anything you don’t understand independently
A number of 98+ students in the sciences point out using multiple sources improves your understanding.
For anyone planning a career in science, that’s also a good habit to get into – if you only have 5 sources on your university research paper, the marker will laugh you out of good marks before they’ve even read your intro.
Research can be pretty threatening, but for HSC level you don’t need to fret hugely.
Some helpful research sources include:
- Using your textbook to verify the accuracy of a source you found on Google (or Google Books!) should be adequate.
- Using HSC Together for FREE video resources that explain each syllabus dot point and Chemistry concepts in detail.
- Using Khan Academy for FREE video resources that explain Chemistry concepts in a visual way in detail.
- Wikipedia (to your teacher’s horror) is generally pretty on point with high school Chemistry.
- If you still feel iffy about Wikipedia, you can look through WIkipedia’s references to find a good source that explores relevant concepts
- Google Scholar can also be used to find primary scientific literature – though some of the papers might be a bit more complex that what is required of you
- Wolfram Alpha is also another database which is basically the Google for math and science
If all the research still left you in the dark, it’s always good to ask your teacher or tutor for advice! With the new syllabus, everyone is in the same position as you so don’t feel afraid that you are the only one needing some navigation.
Struggling with Module 5? Check out our full study guide for Equilibrium and Acid Reactions to build your study notes!
Step 4: Look for connections between syllabus dot points
I know this was stated at the start but it really is an important step to developing a well-rounded understanding of chemistry!
A lot of 6/7 markers in the Chemistry exam can involve information from more than one syllabus point – and this can even extend across multiple modules.
For example, a question can encompass both solution equilibria concepts from Module 5: Equilibrium and Acid Reactions and testing precipitations from Module 8: Applying Chemical Ideas
Try to find any two syllabus points that express high synergy- as there is a good chance that the new HSC exam will question you on both of them at the same time.
To work on our Analysis of Inorganic Substances example, we might need information from:
- 5.4.4: Ksp is an indicator of the solubility of a compound. High Ksp indicates high solubility while low Ksp indicates low solubility.
- 5.4.5: A compound with a lower Q value than its Ksp will not form a precipitate while a compound with a higher Q value than its Ksp will form a precipitate.
- 8.1.2: Precipitation tests is one qualitative way of analysing the presence of a toxic ion in a sample. The Q value when compared to the Ksp of a compound can determine whether it can be detected through a precipitation test. If a compound has lower Q value than Ksp, it cannot be detected through precipitation test and therefore may go undetected.
Now this might seem like a far-fetched question, but the new syllabus likes to test us on our understanding of chemistry concepts so the question might look something like this:
“Explain how Ksp and Q value of a compound may determine the effectiveness of a precipitation test when analyzing inorganic compounds that may be bad for the environment.”
This question might weigh 4 or 5 marks, but notice how it covers the concepts of all three of the syllabus dot points. It isn’t really a route-learning question either as it calls on our conceptual understanding of chemistry.
Step 5: Jump into practice questions
Now you’ve prepped all your HSC Chemistry study notes and they’re top notch, it’s time to test how much you really know!
You can use example questions from textbooks if you want to save HSC papers to practice for the exam.
For practice papers, check out our master list of HSC Chemistry past papers here!
There are plenty of great practice question books you can get, like the Dot Point series, that have questions listed by and centred on each dot point from the syllabus.
To truly test yourself, try filling out the answers in pencil and erasing them in your second attempt to solidify your understanding piece by piece.
If you still want more practice questions, you can also try making your own questions out of the syllabus dot points! Be sure to mark your responses to step into the shoes of your markers and see what they really expect from you!
And that’s it for this article. All the best in your studies!
Trying to write effective study notes for other subjects?
We’ve got guides for a range of subjects, which you can check out below:
- Legal Studies
- Economics
- Visual Arts
- PDHPE
- Standard & Advanced English
- Standard Maths
- Advanced Maths
- Modern History
- Ancient History
- Biology
- Physics
Looking for extra help with HSC Chemistry?
We pride ourselves on our inspirational HSC Chemistry coaches and mentors!
We offer tutoring and mentoring for Years K-12 in a variety of subjects, with personalised lessons conducted one-on-one in your home or at one of our state of the art campuses in Hornsby or the Hills!
To find out more and get started with an inspirational tutor and mentor get in touch today!
Give us a ring on 1300 267 888, email us at [email protected] or check us out on TikTok!
Kate Lynn Law graduated in 2017 with an all rounders HSC award and an ATAR of 97.65. Passionate about mentoring, she enjoys working with high school students to improve their academic, work and life skills in preparation for the HSC and what comes next. An avid blogger, Kate had administrated a creative writing page for over 2000 people since 2013, writing to an international audience since her early teenage years.